
Unit 6: Thermodynamics
Heat and Temperature
Temperature is the average amount of kinetic energy based on molecular motion of a substance
Heat is the flow of energy between two substances at different temperatures
The first law of thermodynamics states energy cannot be created or destroyed and can only transfer or change forms. Energy transfers through molecular collisions.
Exothermic is when energy is transferred from the system to the surroundings, such as when a chemical reaction occurs in a beaker and the beaker feels hot. Endothermic reactions occur when a system absorbs the energy from the surroundings, such as when a beaker feels cold.
Enthalpy
Enthalpy Change, ΔH
Enthalpy change is the measure of energy that is released or absorbed when bonds are broken/formed
When bonds are formed, energy is released. When bonds are broken, energy is absorbed.
If more energy is released than absorbed in a reaction, then it is exothermic and has negative enthalpy change. If more energy is absorbed than released in a reaction, then it is endothermic and has a positive enthalpy change.
Energy Diagrams
Exothermic and Endothermic Reactions
When products have a lower energy than the reactants, the ΔH is negative and it is an exothermic reaction
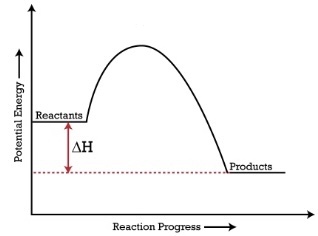
When reactants have a lower energy than the products, the ΔH is positive and it is an endothermic reaction
Catalyst and Energy Diagrams
Catalysts speed up a reaction by lowering the required activation energy and only changes the activation energy, not the amount of energy of products or reactants in the end or beginning (ΔH).
This change can only occur when a reaction is not in equilibrium conditions
Enthalpy of Formation, ΔHf°
Enthalpy of formation, or heat of formation, is the energy change when one mole of a compound is formed from pure elements under standard conditions (298°K, 1 atm)
Example: C(s) + 1/2 O2 (g) + 2 H2 (g) → CH3OH
There is exactly one mole of a product, methanol, which has an odd number of elements, and diatomic in nature.
ΔHf° of a pure element is defined as zero and stands true for elements that are diatomic in pure states, such as hydrogen or oxygen.
If ΔHf° of a compound is negative, energy is released when the compound is formed from pure elements, is exothermic, and is more stable than the reactants.
If ΔHf° is negative, the opposite is true, and it is endothermic.
ΔH can be found if ΔHf° of product and reactants are known
ΣΔHf° Products - ΣΔHf° Reactants = ΔH
The units for reactions enthalpy is kj/mol.
Bond Energy
Bond energy is the energy required to break a bond and is always a positive number. When a bond is formed, the energy of formation is equal to the bond energy released.
ΔH = Σ Bond energy of bond broken - Σ bond energy of bonds formed
Bonds broken are the reactant bonds and the bonds formed are the product bonds
The number of bonds broken is determined by how many moles of that substance there is and how many individual bonds there are.
For 2 H2O molecules, there are two H-O bonds and two of the H2O molecules. So, there are four H-O bonds and to find the bond energy, multiply four by the bond energy of H-O (463 kJ/mol).
Hess’s Law
Hess’s Law states that if a reaction can be broken into a series of steps, the sum of the ΔH of those steps are equal to the ΔH of the reaction.
When manipulating equations for enthalpy calculations…
If a reaction is flipped, flip the enthalpy value (pos to neg, or neg to pos)
If you multiply or divide the coefficients of a reaction, multiply or divide the enthalpy value by that same interval
Adding up the enthalpy values of steps will be the new enthalpy value for the new reaction created from those steps
Enthalpy of Solution
When an ionic substance dissolves in water, energy is absorbed because bonds are breaking, but energy is also emitted because new dipoles are forming.
Step one of dissolving: Bonds are broken in the solute. This step has a positive ΔH because bonds are being broken
Step two: solvent molecules are separated. The water molecules are spread apart to make room for the solvent ions. This weakens intermolecular bonds, resulting in a positive ΔH
Step three: New attractions are created. The free-floating ions are attracted to the dipoles in the water molecules, resulting in a negative ΔH despite no bonds being formed.
Step two and three values are combined to form the hydration energy which is always a negative number. Hydration energy is a Coulombic energy and it’s magnitude increases as the ions increase their charge or decrease the size.
If adding all three steps together, the enthalpy of solution can be determined.
Thermodynamics of Phase Change
Naming the Phase Changes
Solid to liquid: melting
Liquid to solid: freezing
Liquid to gas: vaporization
Gas to liquid: condensation
Solid to gas: sublimation
Gas to solid: deposition.
Vapor pressure is the amount of pressure given off by molecules as they go from solid/liquid to gas.
Enthalpy of Fusion
Enthalpy of fusion is the amount of energy required to cause a solid to melt. Heat of fusion is how much heat given off when a substance freezes. The intermolecular forces in a solid substance are more stable when they are solid so they have a lower energy than liquid forces, so energy is released in the freezing process.
Enthalpy of Vaporization
Enthalpy of vaporization is how much energy is required to turn a liquid to a gas. The heat of vaporization is the energy given off when a gas condenses. Intermolecular forces are more stable in a liquid state, so energy is released as the forces stabilize and get stronger.
When heat is added to a substance, it will either phase change or increase in temperature, not both. Only one can happen at a time. Temperature remains constant during a phase change.
Calorimetry
Calorimetry is the measurement of heat changes during a chemical reaction
The specific heat of substance is how much heat is required to raise one gram of a substance by one degree celsius (or one degree kelvin). The higher or larger a specific heat is, the more heat a substance can absorb without changing temperature. A low specific heat means less heat is required to change the temperature of a substance.
Specific heat equation is q = mcΔT
q is heat added (J or cal)
m is the mass of the substance (g or kg)
c is specific heat
ΔT is temperature change (K or C
Unit 6: Thermodynamics
Heat and Temperature
Temperature is the average amount of kinetic energy based on molecular motion of a substance
Heat is the flow of energy between two substances at different temperatures
The first law of thermodynamics states energy cannot be created or destroyed and can only transfer or change forms. Energy transfers through molecular collisions.
Exothermic is when energy is transferred from the system to the surroundings, such as when a chemical reaction occurs in a beaker and the beaker feels hot. Endothermic reactions occur when a system absorbs the energy from the surroundings, such as when a beaker feels cold.
Enthalpy
Enthalpy Change, ΔH
Enthalpy change is the measure of energy that is released or absorbed when bonds are broken/formed
When bonds are formed, energy is released. When bonds are broken, energy is absorbed.
If more energy is released than absorbed in a reaction, then it is exothermic and has negative enthalpy change. If more energy is absorbed than released in a reaction, then it is endothermic and has a positive enthalpy change.
Energy Diagrams
Exothermic and Endothermic Reactions
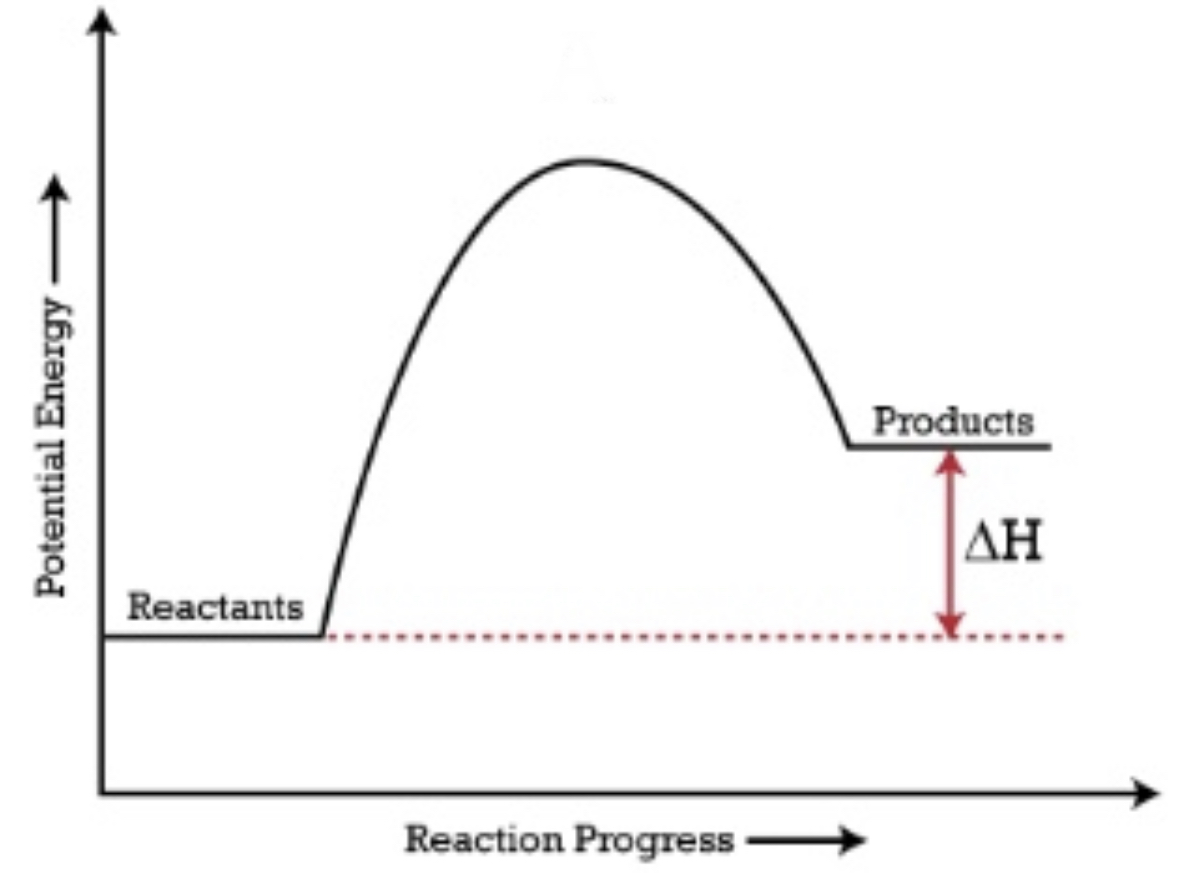
When products have a lower energy than the reactants, the ΔH is negative and it is an exothermic reaction

When reactants have a lower energy than the products, the ΔH is positive and it is an endothermic reaction

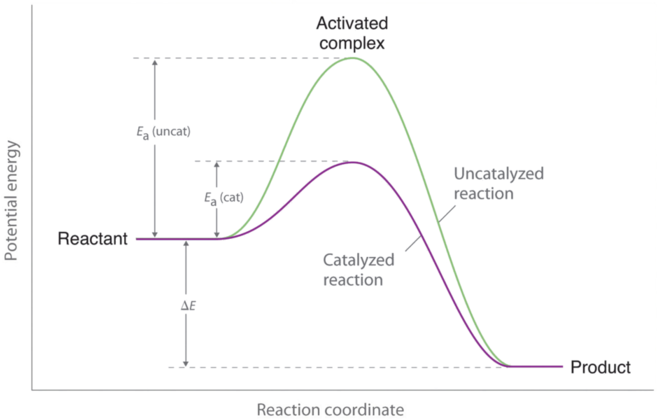
Catalyst and Energy Diagrams

Catalysts speed up a reaction by lowering the required activation energy and only changes the activation energy, not the amount of energy of products or reactants in the end or beginning (ΔH).
This change can only occur when a reaction is not in equilibrium conditions
Enthalpy of Formation, ΔHf°
Enthalpy of formation, or heat of formation, is the energy change when one mole of a compound is formed from pure elements under standard conditions (298°K, 1 atm)
Example: C(s) + 1/2 O2 (g) + 2 H2 (g) → CH3OH
There is exactly one mole of a product, methanol, which has an odd number of elements, and diatomic in nature.
ΔHf° of a pure element is defined as zero and stands true for elements that are diatomic in pure states, such as hydrogen or oxygen.
If ΔHf° of a compound is negative, energy is released when the compound is formed from pure elements, is exothermic, and is more stable than the reactants.
If ΔHf° is negative, the opposite is true, and it is endothermic.
ΔH can be found if ΔHf° of product and reactants are known
ΣΔHf° Products - ΣΔHf° Reactants = ΔH
The units for reactions enthalpy is kj/mol.
Bond Energy
Bond energy is the energy required to break a bond and is always a positive number. When a bond is formed, the energy of formation is equal to the bond energy released.
ΔH = Σ Bond energy of bond broken - Σ bond energy of bonds formed
Bonds broken are the reactant bonds and the bonds formed are the product bonds
The number of bonds broken is determined by how many moles of that substance there is and how many individual bonds there are.
For 2 H2O molecules, there are two H-O bonds and two of the H2O molecules. So, there are four H-O bonds and to find the bond energy, multiply four by the bond energy of H-O (463 kJ/mol).
Hess’s Law
Hess’s Law states that if a reaction can be broken into a series of steps, the sum of the ΔH of those steps are equal to the ΔH of the reaction.
When manipulating equations for enthalpy calculations…
If a reaction is flipped, flip the enthalpy value (pos to neg, or neg to pos)
If you multiply or divide the coefficients of a reaction, multiply or divide the enthalpy value by that same interval
Adding up the enthalpy values of steps will be the new enthalpy value for the new reaction created from those steps
Enthalpy of Solution
When an ionic substance dissolves in water, energy is absorbed because bonds are breaking, but energy is also emitted because new dipoles are forming.
Step one of dissolving: Bonds are broken in the solute. This step has a positive ΔH because bonds are being broken
Step two: solvent molecules are separated. The water molecules are spread apart to make room for the solvent ions. This weakens intermolecular bonds, resulting in a positive ΔH
Step three: New attractions are created. The free-floating ions are attracted to the dipoles in the water molecules, resulting in a negative ΔH despite no bonds being formed.
Step two and three values are combined to form the hydration energy which is always a negative number. Hydration energy is a Coulombic energy and it’s magnitude increases as the ions increase their charge or decrease the size.
If adding all three steps together, the enthalpy of solution can be determined.
Thermodynamics of Phase Change
Naming the Phase Changes
Solid to liquid: melting
Liquid to solid: freezing
Liquid to gas: vaporization
Gas to liquid: condensation
Solid to gas: sublimation
Gas to solid: deposition.
Vapor pressure is the amount of pressure given off by molecules as they go from solid/liquid to gas.
Enthalpy of Fusion
Enthalpy of fusion is the amount of energy required to cause a solid to melt. Heat of fusion is how much heat given off when a substance freezes. The intermolecular forces in a solid substance are more stable when they are solid so they have a lower energy than liquid forces, so energy is released in the freezing process.
Enthalpy of Vaporization
Enthalpy of vaporization is how much energy is required to turn a liquid to a gas. The heat of vaporization is the energy given off when a gas condenses. Intermolecular forces are more stable in a liquid state, so energy is released as the forces stabilize and get stronger.
When heat is added to a substance, it will either phase change or increase in temperature, not both. Only one can happen at a time. Temperature remains constant during a phase change.
Calorimetry
Calorimetry is the measurement of heat changes during a chemical reaction
The specific heat of substance is how much heat is required to raise one gram of a substance by one degree celsius (or one degree kelvin). The higher or larger a specific heat is, the more heat a substance can absorb without changing temperature. A low specific heat means less heat is required to change the temperature of a substance.
Specific heat equation is q = mcΔT
q is heat added (J or cal)
m is the mass of the substance (g or kg)
c is specific heat
ΔT is temperature change (K or C
 Knowt
Knowt
