
Bio
The Building Blocks
Structure
Atom is the smallest unit of matter that has the chemical properties of an element
Atoms contain protons and electrons and neutrons
Exception to this is hydrogen, which has no neutrons
Protons and neutrons have roughly the same mass: one atomic mass unit (amu) or Dalton
Electrons are not considered in atomic mass
Dimer- a molecule consisting of two identical molecules linked together
Most of the volume of an atom (more than 99%) is empty space
Solid objects do not pass through each other because electrons around atoms repel each other
Number and mass
Atomic number is the number of protons
Number of protons and neutrons is the mass number
Atomic mass is the calculated mean of the mass number for its naturally occurring isotopes (often contains fraction)
Isotopes
Different forms of an element with different numbers of neutrons
Some isotopes emit neutrons, protons, and electrons
These are radioactive isotopes, or radioisotopes
Electron shells and the Bohr model
Bohr model shows central nucleus and circular orbitals at specific distances
Octet rule: with the exception of the innermost shell, atoms are more stable energetically when they have 8 electrons in their valence shell
Electrons don’t orbit the nucleus like in Bohr’s model, but are found in electron orbitals
Each electron shell has subshells
Subshells are designated s, p, d, and f with 1, 3, 5, and 7 orbitals respectively
Orbitals can hold two electrons
The nth shell will have n subshells
when adding an electron, it goes to the highest unfilled shell-- when more than one unfilled shells are present, add it to the one that has two fewer subshells filled than the one below it
Subshells are more stable when half full or full
All orbitals will get their first electrons before any get their second
Electron configuration is written as the highest shell, that shell’s subshell, and that subshell’s electrons
For example, Neon (10 electrons) would be written as 1s2 2p2 2p6
Chemical reactions and molecules
Reactions occur when atoms bond or break apart
Substances before the reaction are called the reactants and the substances after the reaction are called the products
An arrow can be used to show a reaction
E.g. 2H2O2 (hydrogen peroxide) → 2H2O (water) + O2 (oxygen)
Molecules with more than one type of element are called compounds
Molecules with one type of element are called homonuclear molecules
Some reactions are reversible
When the concentration of product goes beyond a certain threshold, some of these products will be converted back into reactants
Back and forth continues until equilibrium is reached
This back and forth can be denoted with a double-headed arrow
Law of mass action-- rate of chemical reaction is proportional to the masses of the reacting substances
Ions and ionic bonds
Atoms form ions when they gain or lose electrons to become more stable
Cations are positive ions formed by losing electrons
Anions are negative ions formed by gaining electrons
Anions have their elemental name altered to end in “-ide”
This movement of electrons is called an electron transfer
These can usually only happen simultaneously
Ionic bonds are formed between ions with opposite charges
Covalent bonds and other bonds/interactions
3 pairs of electrons may be shared at most
The more covalent bonds, the stronger the connection between atoms
Covalent bonds are bonds formed when electrons are shared
Metallic bonds
Metallic atom-- has a bunch of electrons on the outer shell that atom is “willing to give”
When many metallic atoms come together, they donate their electrons in a “shared electron pool,” making the atoms positive
The positive atoms become attracted to the negative “electron pool”
Metallic bonds make metals conductive and malleable
London dispersion forces
Weak attractions between atoms or molecules of any kind; depend on temporary imbalances in electron distribution
Because electrons are constantly moving, there will be moments where electrons are clustered together, forming a partial negative charge in one part and a partial positive charge in the other
A molecule with this imbalance can cause a similar charge redistribution in a nearby second molecule, and the molecules will attract because of the temporary charges
Hydrogen bonds and London dispersion forces are both examples of van der Waals forces-- intermolecular interactions that don’t involve covalent bonds or ions
Some textbooks use van der Waals forces only to refer to London dispersion forces
Water
Polarity
The oxygen molecules in water are electronegative, causing a slightly positive charge on hydrogen and slightly negative charge on oxygen
Opposite charges in water form hydrogen bonds
Water attracts/dissolves other polar molecules (hydrophilic) and repels/does not dissolve nonpolar molecules (hydrophobic)
States
Hydrogen bonds give water unique characteristics
Freezing water pushes the molecules apart, making ice less dense than water
Causes a layer of ice to rise to the top of water, which insulates and protects life in the water from freezing
Ice crystals from freezing can rupture membranes
Heat Capacity
Hydrogen bonds cause high specific heat
Specific heat-- amount of heat absorbed/lost for one gram of substance to change one degree celsius
One calorie-- the specific heat of water
Kilocalorie-- 1,000 calories
High heat capacity disperses heat in bodies of animals
Heat of vaporization
Water requires a lot of energy to become a gas due to hydrogen bonds
As a result, water acts as a heat sink
Below the boiling point, individual water molecules with enough energy can still evaporate
Evaporative cooling-- as liquid evaporates, the surface that remains cools; used by many organisms (sweat) to maintain homeostasis of body temperature
Solvent properties
Water’s polarity allows ions and polar molecules to dissolve easily, so it is often referred to as the “solvent of life” or “universal solvent”
Molecules with charge will form hydrogen bonds with and be surrounded by water, forming a sphere of hydration or hydration shell
This keeps molecules separated or dispersed in the water
Reactions with water molecules can disrupt ionic compounds in the process of dissociation (atoms breaking off from molecules to form ions)
Mole - SI unit of an amount of a substance
Molarity - number of moles of solute per liter of solution
Cohesive and adhesive properties
Cohesion-- attraction to molecules of its own kind
Hydrogen bonds allow for surface tension, which causes the formation of droplets and allows things like bugs and paper clips to float on the surface of water
Adhesion- attraction to different molecules
Forms a concave meniscus on glass
Observed when water “climbs” a thin glass capillary tube
This “climbing” is called capillary action
Transports water from roots to rest of plant
Insects stay afloat on water because of surface tension
Buffers, acids, and bases
pH
Litmus tests acidity or alkalinity
In pure water, when water becomes hydrogen ions and hydroxide ions, hydroxide stays bound to other molecules because of hydrogen bonds and hydrogen ions form H3O with other water molecules
Negative of the base 10 logarithm of the concentration of hydrogen ions is the pH
Near-neutral pH is maintained in blood and in human cells
Acids and bases
Acids increase the concentration of hydrogen ions (lower pH) usually by having one of its own hydrogen ions dissociate
bases lower the concentration of hydrogen ions (higher pH) by providing hydroxide ions or another anion to combine with hydrogen ions
Stronger acids more readily give hydrogen ions, like hydrochloric acid, which dissociates into hydrogen and chloride ions, unlike tomato juice and vinegar do not completely dissociate and are weak acids
Strong bases readily take up hydrogen ions, like sodium hydroxide and many other household cleaners that rapidly give up hydroxide ions, unlike sea water, with a pH near 8.0
Lower than 7 on the pH scale is acidic and higher than 7 on the pH scale is alkaline
High acidity in stomach causes cells to constantly be replaced in the stomach
Buffers
Buffers absorb excess hydrogen ions or hydroxide ions to maintain a near neutral pH
Buffer in human blood involves carbonic acid (H2CO3), bicarbonate ion (HCO3–), and carbon dioxide (CO2)
Bicarbonate combines with hydrogen ions and become carbonic acid
Carbonic acid becomes water and carbon dioxide, and carbon dioxide is exhaled
Carbonic acid can combine with excess dioxide ions to make bicarbonate
Carbon
Hydrocarbons
Organic molecules made entirely of carbon and hydrogen
Covalent bonds between the atoms store a lot of energy, allowing it to be used as fuel when burned (oxidized) e.g. methane (CH4)
May exist as linear chains, rings, or combinations of both
May be single, double, or triple bonds
Isomers
Isomers are molecules that share the same chemical formula but differ in the placement of their atoms and/or chemical bonds
Structural isomers differ in the placement of their covalent bonds
Geometric isomers differ in how covalent bonds are made to the surrounding atoms, especially carbon-to-carbon double bonds
Same side of a double bond is a cis configuration
Opposite sides of a double bond is a trans configuration
Enantiomers
Enantiomers are molecules that share the same chemical structure and chemical bonds but differ in the three-dimensional placement of atoms so that they are mirror images (e.g. below)
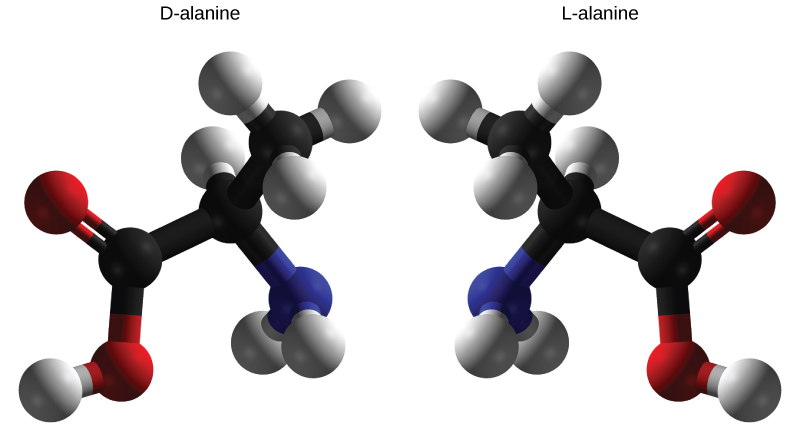
Stereoisomers
Stereoisomers are molecules that share the same chemical structure and bonds but have a different organization of atoms around one carbon
Functional Groups
Functional groups are groups of atoms that occur within molecules and confer specific chemical properties to those molecules
Found along the “carbon backbone” of macromolecules-- chains/rings of carbon atoms with occasional substitutions of elements like nitrogen or oxygen
These molecules with other elements in their carbon backbone are substituted hydrocarbons
Functional groups can participate in specific chemical reactions
Examples
Hydroxyl (R-O-H | Polar)
Methyl (R-CH3 | Nonpolar)
Carbonyl (R-C*-R’ *--O | Polar)
Carboxyl (O--C*-OH *-R | charged and ionized to release hydrogen ions)
Amino (H-N*-H *-R | takes hydrogen ions to form NH3+)
Hydrogen bonds are important in folding properly into and maintaining the shape for functioning
Synthesis of Biological Macromolecules
Condensation reaction - molecules covalently bond through the loss of a small molecule
Dehydration synthesis - condensation reaction where a water molecule is lost
Biological macromolecules-- large molecules necessary for life that built from smaller organic molecules
Four major classes of biological macromolecules
Carbohydrates
Lipids
Proteins
Nucleic acids
Most macromolecules made from single subunits called monomers
Monomers combine with each other using covalent bonds to form larger molecules called polymers, releasing water molecules as byproducts
This reaction is known as dehydration synthesis
In this reaction, the hydrogen of one monomer combines with the hydroxyl group of another monomer, releasing a molecule of water
e.g. an electronegative oxygen in a glucose molecule may bond with an electropositive carbon atom in another glucose molecule; the hydrogen bonded to the oxygen may separate and the hydroxyl bonded to the carbon may separate, and the two may combine to make water
Alternatively, a water molecule may pick up the oxygen’s hydrogen atom and form a hydronium ion (H3O+)
Hydrolysis
Polymers break into monomers through hydrolysis
Hydrolysis is a reaction in which a water molecule is used to break down another compound
The polymer is broken into two components, one that gains a hydrogen atom and one that gains a hydroxyl molecule from a split water molecule
Specific enzymes catalyze (speed up) dehydration and hydrolysis reactions
Dehydration reactions form new bonds and requires energy while hydrolysis reactions break bonds and release energy
Carbohydrates
Molecular Structures
Carbohydrates provide energy to the body, particularly through glucose, a simple sugar that is a component of starch
Carbohydrates can be represented by the stoichiometric formula (CH2O)n where n is the number of carbons in the molecule
Classified into 3 subtypes
Monosaccharides
Monosaccharides are simple sugars; the most common is glucose
Usually has 3-7 carbons
Most monosaccharide names end with “-ose”
If it has an aldehyde group (R-C*-H *--O, i.e. it has its carbonyl group on the end) it is known as an aldose
If it has a ketone group (R-C*-R *--O i.e. it has its carbonyl group internally) it is known as a ketose
Depending on the number of carbons, they may also be known as trioses (3), pentoses (5), or hexoses (6)
Chemical formula for glucose (used as energy by humans and plants), galactose (part of milk sugar) and fructose (found in fruit) is C6H12O6, these all have the same chemical formula but differ structurally and chemically from each other
These 3 monosaccharides are isomeric hexoses (6 carbons and different structures)
Glucose and galactose are aldoses while fructose is a ketose
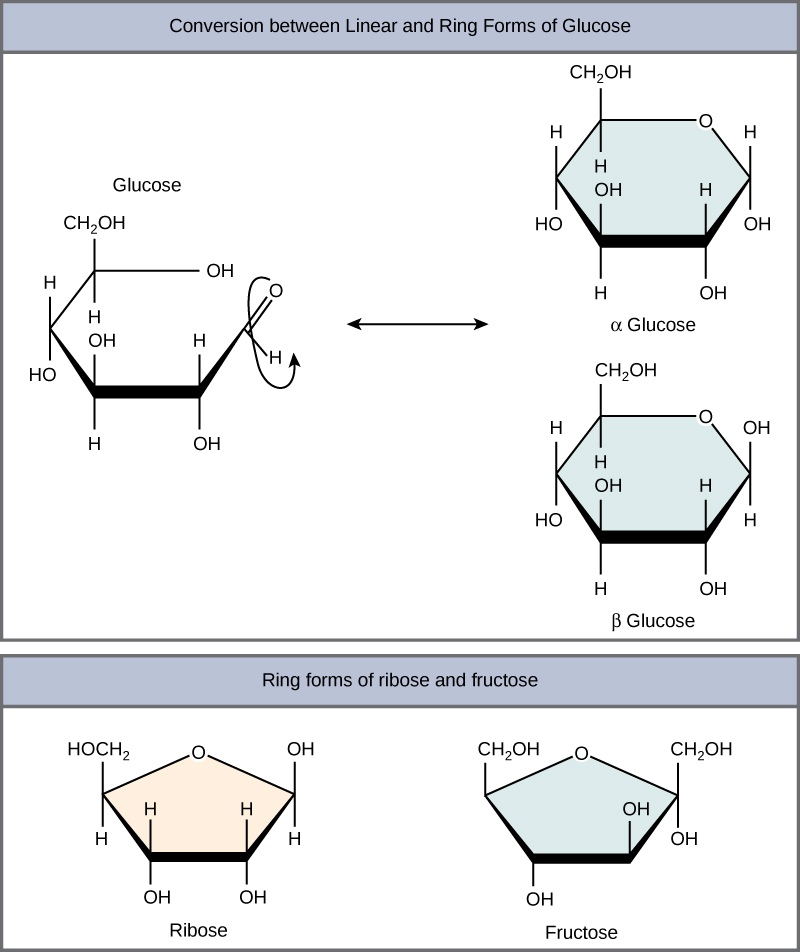
Monosaccharides in aqueous solutions are usually found in ring forms
Glucose in a ring form can have two different arrangements of the hydroxyl group around the anomeric carbon (carbon that becomes asymmetric in the process of ring formation)
If the hydroxyl group is below the anomeric carbon/plane, it is in the alpha (a) position
If the hydroxyl group is above, it is in the beta (b**)** position (image for reference)
Disaccharides
Form when two monosaccharides undergo a dehydration reaction
The hydroxyl group of one monosaccharide combines with the hydrogen of another, releasing a water molecule and forming a covalent bond known as a glycosidic bond, which can be the alpha or beta type
A glycosidic bond is an oxygen bonded to the carbons of two sugars
Common disaccharides include lactose (found in milk), maltose (formed by dehydration synthesis between two glucose molecules) and sucrose (table sugar), the most common and composed of glucose and fructose
Polysaccharides
Polysaccharides are long chains of monosaccharides linked by glycosidic bonds
Chain may be branched or unbranched, may contain different types of monosaccharides
Primary examples include starch, glycogen, cellulose, and chitin
Starch
Stored form of sugars in plants made of a mixture of amylose (linear chain structure) and amylopectin (branch chain structure), two polymers of glucose
Plants can synthesize glucose, and the excess glucose is stored as starch in different plant parts including the roots and the seeds
The starch in the seeds provides food for the embryo as it germinates and can also act as a food source for humans and animals
Starch consumed by humans is broken down by enzymes into smaller molecules like glucose and maltose, which is then absorbed by the cells
Made of glucose monomers joined by a 1-4 or a 1-6 bonds glycosidic bonds
The numbers refer to the carbon number of the two residues that join to form the bond (right-most carbon is 1, counting clockwise, left-most is 4)
Glycogen
Glycogen is the storage form of glucose in humans and other vertebrates and is made of glucose monomers
Glycogen is the animal equivalent of starch and is a highly branched molecule usually stored in liver and muscle cells
When glucose levels in the blood decrease, glycogen is broken down to release glucose in a process called glycogenolysis
Cellulose
The most abundant natural biopolymer
Cell wall of plants is mostly made of cellulose; provides structural support to the cell
Wood and paper are mostly cellulosic in nature
Made of glucose monomers linked by b 1-4 glycosidic bonds
Every other glucose monomer in cellulose is flipped over and the monomers are packed tightly as extended long chains, giving cellulose rigidity and strength, which is important to plant cells
The b 1-4 linkage can’t be broken down by human digestive enzymes, but herbivores like cows and buffalos can with the help of specialized flora in their stomach to digest plant material rich in cellulose
Certain species of bacteria and protists reside in the rumen (part of the digestive system of herbivores) and secrete the enzyme cellulase
The appendix also contains bacteria that digest cellulose
Cellulases break down cellulose into glucose monomers to be used for energy
Termites can also break down cellulose because of other organisms in their body that secrete cellulases
Chitin
A polysaccharide-containing nitrogen
Composes the exoskeleton of arthropods to protect internal body parts
Made of repeating units of a modified sugar called N-acetyl-b-d-glucosamine
Also a major component of fungal cell walls
Lipids
Sources of energy that power cellular processes, usually nonpolar and hydrophobic
Lipids are usually nonpolar because they are hydrocarbons that include mostly carbon-carbon/hydrogen-carbon bonds
Fats and Oils
Purposes
Cells store energy in the form of fats
Lipids provide insulation, e.g. birds and mammals can keep dry with a protective layer over fur or feathers
Lipids are the building blocks of many hormones and important in constructing cellular membranes
Lipids include fats, waxes, phospholipids, and steroids
Structure
A fat molecule consists of mainly glycerol and fatty acids
Glycerol is an organic compound (alcohol) with 3 carbons, 5 hydrogens, and 3 hydroxyl groups
Fatty acids have long chains of hydrocarbons attached to a carboxyl group
In a fat molecule, fatty acids are attached the glycerol’s carbons by dehydration synthesis with ester bonds through oxygen atoms
Ester bonds - a carbon with a single bond to a carbon, a double bond to an oxygen, and a single bond to an oxygen bonded to another carbon (R-O-C*--O *-R)
Fats are also called triacylglycerols/triglycerides because of their structure
They have “acyl” in them because the (O--C-C) group connecting the glycerol to the hydrocarbon chain is called an acyl group
Fatty acids are saturated if carbons have single bonds (“saturated” with hydrogen) while unsaturated fatty acids have double bonds
Oils
Unsaturated fats are typically liquid at room temperature and are called oils
One double bond in the molecule is monounsaturated and more than one double bond is polyunsaturated
Fats are only saturated when there are no double bonds
Unsaturated fats and oils contain cis fatty acids
Cis - hydrogens are in the same plane (causes a bend that prevents the acids from packing tightly, keeping them liquid at room temperature)
Trans - hydrogens are on two different planes
Trans Fats
Trans fats are artificially hydrogenated to make them semi-solid and have a more desirable consistency
Hydrogen gas is bubbled through oils which may double bonds from cis to trans
Omega Fatty Acids
Fatty acids not required but not synthesized by the human body are essential fatty acids and must be supplemented through the diet
The two known essential fatty acids are omega-3 and omega-6
omega-3 is polyunsaturated and the third carbon from the omega (w) carbon is connected to the fourth by a double bond
Good sources of omega-3 are fish like salmon and tuna
The furthest carbon away from the carboxyl group is numbered as the omega (w) carbon
Waxes
Made of long fatty acid chains esterified to long-chain alcohols
Because it’s hydrophobic, it covers some feathers and some leaves to prevent water from sticking to the surface
Phospholipids
Major constituents of the plasma membrane (outermost layer of living cells)
Composed of fatty acid chains attached to a glycerol or sphingosine backbone
Differs from fats because there are only two fatty acids forming a diacylglycerol, the third carbon is occupied by a modified phosphate group
A phosphate group attached to a diacylglycerol is a phosphatidate and becomes a phospholipid when modified by an alcohol
Phospholipids are amphipathic, meaning it has a hydrophilic and a hydrophobic part (the phosphate head is hydrophilic and the lipid tail contains the hydrophobic fatty acids)
phospholipids placed in water form a micelle, where the heads face the outside and the fatty acids face the interior
Steroids
Steroids have a fused ring structure
Hydrophobic and insoluble in water
Steroids all have four linked carbon rings and several have a short tail (like cholesterol)
Many steroids also have an -OH functional group, putting them in the alcohol classification
Most common steroid is cholesterol, which is mainly synthesized in the liver and is used for many things like testosterone and Vitamin D
Proteins
Proteins are long chains of different sequences of the 20 amino acids that each contain an amino group (-NH2), a carboxyl group (OH-C--O), and a variable group
Amino acids are linked by a peptide bond formed through a dehydration reaction; a long chain of amino acids is a polypeptide
Changes in temperature, pH, and exposure to chemicals may change the protein, making it lose its function (denaturation)
Types and functions
Proteins have the most diverse range of functions of all macromolecules and their structures vary greatly, although they are all polymers of amino acids in a linear sequence
Enzymes
Produced by living cells and are catalysts in biochemical reactions like digestion
Speeds up specific reactions by decreasing the amount of energy needed
Each enzyme is specific for the substrate (a reactant that binds to the enzyme) it acts on and may help in breakdown, rearrangement, or synthesis reactions
Enzymes that break down their substrate are catabolic, enzymes that build more complex molecules are anabolic, and enzymes that affect the rate of reaction are catalytic
All enzymes increase the rate of reaction and so are organic catalysts
Hormones
Chemical-signaling molecules, usually small proteins or steroids, secreted by endocrine cells that act to control specific processes
Primary functions
Digestive enzymes - catabolize nutrients into monomeric units
Transport - carry substances in the blood or lymph
Structural - make different structures e.g. cytoskeleton
Hormones - coordinate activities
Defense - protect from foreign pathogens
Contractile - effect muscle contraction
Storage - nourish the embryo/seedling during early development
Amino acids
Amino acids have a central alpha (a) carbon bonded to an amino group (NH2), a carboxyl group (COOH), a side chain, and a hydrogen atom
The side chain determines the nature of the amino acid (pH, polar/nonpolar)
Amino acids are represented by a single uppercase letter or a three-letter abbreviation (Valine - V or val)
Essential amino acids are necessary for proteins but not produced by the body
The sequence and number of amino acids determine the shape, size, and function of the protein
Amino acids are attached by a covalent bond known as a peptide bond, which is formed by a dehydration reaction
Linked amino acids form peptides, and multiple amino acids linked form a polypeptide with an amino group at one end (N or the amino terminal) and a carboxyl group at the other (C or carboxyl terminal)
After protein synthesis, proteins are modified through post-translational modifications that make it completely functional
The carboxyl group will lose its hydrogen proton and the amino group will have gained a hydrogen proton
A neutral molecule where parts have charges that balance each other out is called a zwitterion
Protein Structure
Enzymes bind to a specific substrate at a site known as the active site (region of enzyme surface where substrate binds)
Primary structure
The sequence of amino acids in a polypeptide chain is its primary structure
The sequence is determined by the gene encoding the protein; a change in the nucleotide sequence of the gene’s coding may affect protein structure and function
Secondary structure
The folding of the polypeptide in some regions is the secondary structure
Formed by hydrogen bonds between amino hydrogen and carboxyl oxygen on the backbones
The most common are the a-helix (spirally) and B-pleated sheet (zig-zaggy) structures, both of which are held by hydrogen bonds
Tertiary structure
The three-dimensional structure of a polypeptide is its tertiary structure
This structure is partly because of chemical interactions on the chains, primarily interactions between R groups
These interactions can counteract hydrogen bonds in standard secondary structures
Produced by hydrophobic interactions, ionic bonds, covalent bonds, and van der Waals forces
Quaternary structure
Interactions between subunits of polypeptides form the quaternary structure
Weak interactions between subunits stabilize the overall structure
Denaturation and protein folding
Temperature, pH, and chemicals may change the protein structure
When proteins lose their shape without losing their primary sequence, it is known as denaturation
Denaturation is often reversible because the primary structure of the polypeptide is conserved if the denaturing agent is removed
Chaperones
Protein helpers that assist during the folding process
They associate with the target protein and prevent aggregation of polypeptides that make up the complete protein structure
they disassociate from the protein after the process
Folding is critical to protein process
Nucleic Acids
DNA and RNA
Nucleic acids carry the genetic blueprint of a cell and instructions for cell functions
Two main types
Deoxyribonucleic acid (DNA)
Ribonucleic acid (RNA)
Genes
Genes may carry information to make protein or RNA products
Genome - the genetic content of a cell
Genomics - the study of genomes
DNA forms a complex with histone proteins to form chromatin - the substance of eukaryotic chromosomes, which may contain thousands of genes
DNA controls genes by turning them “on” or “off”
RNA
RNA is mostly involved with protein synthesis and regulation
DNA molecules never leave the nucleus, so messenger RNA (mRNA) are used to communicate with the rest of the sell
DNA and RNA
Made up of monomers known as nucleotides, which combine to form polynucleotides
Nucleotides have three parts
A nitrogenous base
Organic molecules (they contain nitrogen and carbon)
They are bases because they contain an amino group
Four possible bases
adenine (A) and guanine (G)
Classified as purines
Primary structure of a purine is two carbon-nitrogen rings
Cytosine (C), Uracil (U), and Thymine (T)
Pyrimidines
Primary structure is a single carbon-nitrogen ring
Uracil replaces Thymine in RNA; they are very similar, but thymine has a methyl group
A pentose sugar
Deoxyribose in DNA and ribose in RNA
On the second carbon (2’), ribose has a hydroxyl group and deoxyribose has a hydrogen
A phosphate group
Phosphate residue is attached to the hydroxyl groups of the 5’ carbon of one sugar and the 3’ carbon of the sugar of the next nucleotide, forming a 5’-3’ phosphodiester linkage
Not formed by dehydration reaction like others
Formed through the removal of two phosphate groups
DNA Double-Helix Structure
Sugars and phosphate lie on the outside of the helix, forming the DNA’s backbone
Nitrogenous bases are stacked on the interior in pairs; pairs are bound by hydrogen bonds
The two strands will run in opposite directions, making the 5’ carbon of one strand face the 3’ carbon of the other
This is called antiparallel orientation
It is important to DNA replication and nucleic acid interactions
Base complementary rule - A pairs with T and G pairs with C; this is due to the way the hydrogen bonds form between the nitrogenous bases
During DNA replication, each strand is copied, so the daughter DNA double helix contains one parental DNA strand and one newly synthesized strand
RNA
RNA is usually single-stranded and made of ribonucleotides linked by phosphodiester bonds
A ribonucleotide contains ribose, a nitrogenous base, and the phosphate group
Four main types of RNA
Messenger RNA (mRNA)
Carries messages from the DNA
Base sequence is complementary to the sequence of the DNA it was copied from, but U replaces T
Interacts with ribosomes and other machinery in the cytoplasm
Read in sets of three bases known as codons
Each codon codes for a single amino acid
This allows the mRNA to be read and produce a protein product
Ribosomal RNA (rRNA)
A major constituent of the ribosomes mRNA bind on
Ensures proper alignment of the mRNA and ribosomes
Catalyzes the formation of peptide bonds between two amino acids
Transfer RNA (tRNA)
One of the smallest of the four types
Carries the correct amino acid to the site of protein synthesis
The base pairing between tRNA and mRNA allows for the correct amino acid to be inserted into the polypeptide chain
tRNA has anticodons that pair with the codons on the mRNA
microRNA
Smallest RNA molecules
Regulate gene expression by interfering with the expression of certain mRNA messages
DNA and RNA comparison
Features of DNA and RNA |
|---|
Function |
Location |
Structure |
Sugar |
Pyrimidines |
Purines |
DNA dictates the structure of mRNA in a transcription process, and RNA dictates the structure of protein in a translation process
Bio
The Building Blocks
Structure
Atom is the smallest unit of matter that has the chemical properties of an element
Atoms contain protons and electrons and neutrons
Exception to this is hydrogen, which has no neutrons
Protons and neutrons have roughly the same mass: one atomic mass unit (amu) or Dalton
Electrons are not considered in atomic mass
Dimer- a molecule consisting of two identical molecules linked together
Most of the volume of an atom (more than 99%) is empty space
Solid objects do not pass through each other because electrons around atoms repel each other
Number and mass
Atomic number is the number of protons
Number of protons and neutrons is the mass number
Atomic mass is the calculated mean of the mass number for its naturally occurring isotopes (often contains fraction)
Isotopes
Different forms of an element with different numbers of neutrons
Some isotopes emit neutrons, protons, and electrons
These are radioactive isotopes, or radioisotopes
Electron shells and the Bohr model
Bohr model shows central nucleus and circular orbitals at specific distances
Octet rule: with the exception of the innermost shell, atoms are more stable energetically when they have 8 electrons in their valence shell
Electrons don’t orbit the nucleus like in Bohr’s model, but are found in electron orbitals
Each electron shell has subshells
Subshells are designated s, p, d, and f with 1, 3, 5, and 7 orbitals respectively
Orbitals can hold two electrons
The nth shell will have n subshells
when adding an electron, it goes to the highest unfilled shell-- when more than one unfilled shells are present, add it to the one that has two fewer subshells filled than the one below it
Subshells are more stable when half full or full
All orbitals will get their first electrons before any get their second
Electron configuration is written as the highest shell, that shell’s subshell, and that subshell’s electrons
For example, Neon (10 electrons) would be written as 1s2 2p2 2p6
Chemical reactions and molecules
Reactions occur when atoms bond or break apart
Substances before the reaction are called the reactants and the substances after the reaction are called the products
An arrow can be used to show a reaction
E.g. 2H2O2 (hydrogen peroxide) → 2H2O (water) + O2 (oxygen)
Molecules with more than one type of element are called compounds
Molecules with one type of element are called homonuclear molecules
Some reactions are reversible
When the concentration of product goes beyond a certain threshold, some of these products will be converted back into reactants
Back and forth continues until equilibrium is reached
This back and forth can be denoted with a double-headed arrow
Law of mass action-- rate of chemical reaction is proportional to the masses of the reacting substances
Ions and ionic bonds
Atoms form ions when they gain or lose electrons to become more stable
Cations are positive ions formed by losing electrons
Anions are negative ions formed by gaining electrons
Anions have their elemental name altered to end in “-ide”
This movement of electrons is called an electron transfer
These can usually only happen simultaneously
Ionic bonds are formed between ions with opposite charges
Covalent bonds and other bonds/interactions
3 pairs of electrons may be shared at most
The more covalent bonds, the stronger the connection between atoms
Covalent bonds are bonds formed when electrons are shared
Metallic bonds
Metallic atom-- has a bunch of electrons on the outer shell that atom is “willing to give”
When many metallic atoms come together, they donate their electrons in a “shared electron pool,” making the atoms positive
The positive atoms become attracted to the negative “electron pool”
Metallic bonds make metals conductive and malleable
London dispersion forces
Weak attractions between atoms or molecules of any kind; depend on temporary imbalances in electron distribution
Because electrons are constantly moving, there will be moments where electrons are clustered together, forming a partial negative charge in one part and a partial positive charge in the other
A molecule with this imbalance can cause a similar charge redistribution in a nearby second molecule, and the molecules will attract because of the temporary charges
Hydrogen bonds and London dispersion forces are both examples of van der Waals forces-- intermolecular interactions that don’t involve covalent bonds or ions
Some textbooks use van der Waals forces only to refer to London dispersion forces
Water
Polarity
The oxygen molecules in water are electronegative, causing a slightly positive charge on hydrogen and slightly negative charge on oxygen
Opposite charges in water form hydrogen bonds
Water attracts/dissolves other polar molecules (hydrophilic) and repels/does not dissolve nonpolar molecules (hydrophobic)
States
Hydrogen bonds give water unique characteristics
Freezing water pushes the molecules apart, making ice less dense than water
Causes a layer of ice to rise to the top of water, which insulates and protects life in the water from freezing
Ice crystals from freezing can rupture membranes
Heat Capacity
Hydrogen bonds cause high specific heat
Specific heat-- amount of heat absorbed/lost for one gram of substance to change one degree celsius
One calorie-- the specific heat of water
Kilocalorie-- 1,000 calories
High heat capacity disperses heat in bodies of animals
Heat of vaporization
Water requires a lot of energy to become a gas due to hydrogen bonds
As a result, water acts as a heat sink
Below the boiling point, individual water molecules with enough energy can still evaporate
Evaporative cooling-- as liquid evaporates, the surface that remains cools; used by many organisms (sweat) to maintain homeostasis of body temperature
Solvent properties
Water’s polarity allows ions and polar molecules to dissolve easily, so it is often referred to as the “solvent of life” or “universal solvent”
Molecules with charge will form hydrogen bonds with and be surrounded by water, forming a sphere of hydration or hydration shell
This keeps molecules separated or dispersed in the water
Reactions with water molecules can disrupt ionic compounds in the process of dissociation (atoms breaking off from molecules to form ions)
Mole - SI unit of an amount of a substance
Molarity - number of moles of solute per liter of solution
Cohesive and adhesive properties
Cohesion-- attraction to molecules of its own kind
Hydrogen bonds allow for surface tension, which causes the formation of droplets and allows things like bugs and paper clips to float on the surface of water
Adhesion- attraction to different molecules
Forms a concave meniscus on glass
Observed when water “climbs” a thin glass capillary tube
This “climbing” is called capillary action
Transports water from roots to rest of plant
Insects stay afloat on water because of surface tension
Buffers, acids, and bases
pH
Litmus tests acidity or alkalinity
In pure water, when water becomes hydrogen ions and hydroxide ions, hydroxide stays bound to other molecules because of hydrogen bonds and hydrogen ions form H3O with other water molecules
Negative of the base 10 logarithm of the concentration of hydrogen ions is the pH
Near-neutral pH is maintained in blood and in human cells
Acids and bases
Acids increase the concentration of hydrogen ions (lower pH) usually by having one of its own hydrogen ions dissociate
bases lower the concentration of hydrogen ions (higher pH) by providing hydroxide ions or another anion to combine with hydrogen ions
Stronger acids more readily give hydrogen ions, like hydrochloric acid, which dissociates into hydrogen and chloride ions, unlike tomato juice and vinegar do not completely dissociate and are weak acids
Strong bases readily take up hydrogen ions, like sodium hydroxide and many other household cleaners that rapidly give up hydroxide ions, unlike sea water, with a pH near 8.0
Lower than 7 on the pH scale is acidic and higher than 7 on the pH scale is alkaline
High acidity in stomach causes cells to constantly be replaced in the stomach
Buffers
Buffers absorb excess hydrogen ions or hydroxide ions to maintain a near neutral pH
Buffer in human blood involves carbonic acid (H2CO3), bicarbonate ion (HCO3–), and carbon dioxide (CO2)
Bicarbonate combines with hydrogen ions and become carbonic acid
Carbonic acid becomes water and carbon dioxide, and carbon dioxide is exhaled
Carbonic acid can combine with excess dioxide ions to make bicarbonate
Carbon
Hydrocarbons
Organic molecules made entirely of carbon and hydrogen
Covalent bonds between the atoms store a lot of energy, allowing it to be used as fuel when burned (oxidized) e.g. methane (CH4)
May exist as linear chains, rings, or combinations of both
May be single, double, or triple bonds
Isomers
Isomers are molecules that share the same chemical formula but differ in the placement of their atoms and/or chemical bonds
Structural isomers differ in the placement of their covalent bonds
Geometric isomers differ in how covalent bonds are made to the surrounding atoms, especially carbon-to-carbon double bonds
Same side of a double bond is a cis configuration
Opposite sides of a double bond is a trans configuration
Enantiomers
Enantiomers are molecules that share the same chemical structure and chemical bonds but differ in the three-dimensional placement of atoms so that they are mirror images (e.g. below)

Stereoisomers
Stereoisomers are molecules that share the same chemical structure and bonds but have a different organization of atoms around one carbon
Functional Groups
Functional groups are groups of atoms that occur within molecules and confer specific chemical properties to those molecules
Found along the “carbon backbone” of macromolecules-- chains/rings of carbon atoms with occasional substitutions of elements like nitrogen or oxygen
These molecules with other elements in their carbon backbone are substituted hydrocarbons
Functional groups can participate in specific chemical reactions
Examples
Hydroxyl (R-O-H | Polar)
Methyl (R-CH3 | Nonpolar)
Carbonyl (R-C*-R’ *--O | Polar)
Carboxyl (O--C*-OH *-R | charged and ionized to release hydrogen ions)
Amino (H-N*-H *-R | takes hydrogen ions to form NH3+)
Hydrogen bonds are important in folding properly into and maintaining the shape for functioning
Synthesis of Biological Macromolecules
Condensation reaction - molecules covalently bond through the loss of a small molecule
Dehydration synthesis - condensation reaction where a water molecule is lost
Biological macromolecules-- large molecules necessary for life that built from smaller organic molecules
Four major classes of biological macromolecules
Carbohydrates
Lipids
Proteins
Nucleic acids
Most macromolecules made from single subunits called monomers
Monomers combine with each other using covalent bonds to form larger molecules called polymers, releasing water molecules as byproducts
This reaction is known as dehydration synthesis
In this reaction, the hydrogen of one monomer combines with the hydroxyl group of another monomer, releasing a molecule of water
e.g. an electronegative oxygen in a glucose molecule may bond with an electropositive carbon atom in another glucose molecule; the hydrogen bonded to the oxygen may separate and the hydroxyl bonded to the carbon may separate, and the two may combine to make water
Alternatively, a water molecule may pick up the oxygen’s hydrogen atom and form a hydronium ion (H3O+)
Hydrolysis
Polymers break into monomers through hydrolysis
Hydrolysis is a reaction in which a water molecule is used to break down another compound
The polymer is broken into two components, one that gains a hydrogen atom and one that gains a hydroxyl molecule from a split water molecule
Specific enzymes catalyze (speed up) dehydration and hydrolysis reactions
Dehydration reactions form new bonds and requires energy while hydrolysis reactions break bonds and release energy
Carbohydrates
Molecular Structures
Carbohydrates provide energy to the body, particularly through glucose, a simple sugar that is a component of starch
Carbohydrates can be represented by the stoichiometric formula (CH2O)n where n is the number of carbons in the molecule
Classified into 3 subtypes
Monosaccharides
Monosaccharides are simple sugars; the most common is glucose
Usually has 3-7 carbons
Most monosaccharide names end with “-ose”
If it has an aldehyde group (R-C*-H *--O, i.e. it has its carbonyl group on the end) it is known as an aldose
If it has a ketone group (R-C*-R *--O i.e. it has its carbonyl group internally) it is known as a ketose
Depending on the number of carbons, they may also be known as trioses (3), pentoses (5), or hexoses (6)
Chemical formula for glucose (used as energy by humans and plants), galactose (part of milk sugar) and fructose (found in fruit) is C6H12O6, these all have the same chemical formula but differ structurally and chemically from each other
These 3 monosaccharides are isomeric hexoses (6 carbons and different structures)
Glucose and galactose are aldoses while fructose is a ketose
Monosaccharides in aqueous solutions are usually found in ring forms
Glucose in a ring form can have two different arrangements of the hydroxyl group around the anomeric carbon (carbon that becomes asymmetric in the process of ring formation)
If the hydroxyl group is below the anomeric carbon/plane, it is in the alpha (a) position
If the hydroxyl group is above, it is in the beta (b**)** position (image for reference)
Disaccharides

Form when two monosaccharides undergo a dehydration reaction
The hydroxyl group of one monosaccharide combines with the hydrogen of another, releasing a water molecule and forming a covalent bond known as a glycosidic bond, which can be the alpha or beta type
A glycosidic bond is an oxygen bonded to the carbons of two sugars
Common disaccharides include lactose (found in milk), maltose (formed by dehydration synthesis between two glucose molecules) and sucrose (table sugar), the most common and composed of glucose and fructose
Polysaccharides
Polysaccharides are long chains of monosaccharides linked by glycosidic bonds
Chain may be branched or unbranched, may contain different types of monosaccharides
Primary examples include starch, glycogen, cellulose, and chitin
Starch
Stored form of sugars in plants made of a mixture of amylose (linear chain structure) and amylopectin (branch chain structure), two polymers of glucose
Plants can synthesize glucose, and the excess glucose is stored as starch in different plant parts including the roots and the seeds
The starch in the seeds provides food for the embryo as it germinates and can also act as a food source for humans and animals
Starch consumed by humans is broken down by enzymes into smaller molecules like glucose and maltose, which is then absorbed by the cells
Made of glucose monomers joined by a 1-4 or a 1-6 bonds glycosidic bonds
The numbers refer to the carbon number of the two residues that join to form the bond (right-most carbon is 1, counting clockwise, left-most is 4)
Glycogen
Glycogen is the storage form of glucose in humans and other vertebrates and is made of glucose monomers
Glycogen is the animal equivalent of starch and is a highly branched molecule usually stored in liver and muscle cells
When glucose levels in the blood decrease, glycogen is broken down to release glucose in a process called glycogenolysis
Cellulose
The most abundant natural biopolymer
Cell wall of plants is mostly made of cellulose; provides structural support to the cell
Wood and paper are mostly cellulosic in nature
Made of glucose monomers linked by b 1-4 glycosidic bonds
Every other glucose monomer in cellulose is flipped over and the monomers are packed tightly as extended long chains, giving cellulose rigidity and strength, which is important to plant cells
The b 1-4 linkage can’t be broken down by human digestive enzymes, but herbivores like cows and buffalos can with the help of specialized flora in their stomach to digest plant material rich in cellulose
Certain species of bacteria and protists reside in the rumen (part of the digestive system of herbivores) and secrete the enzyme cellulase
The appendix also contains bacteria that digest cellulose
Cellulases break down cellulose into glucose monomers to be used for energy
Termites can also break down cellulose because of other organisms in their body that secrete cellulases
Chitin
A polysaccharide-containing nitrogen
Composes the exoskeleton of arthropods to protect internal body parts
Made of repeating units of a modified sugar called N-acetyl-b-d-glucosamine
Also a major component of fungal cell walls
Lipids
Sources of energy that power cellular processes, usually nonpolar and hydrophobic
Lipids are usually nonpolar because they are hydrocarbons that include mostly carbon-carbon/hydrogen-carbon bonds
Fats and Oils
Purposes
Cells store energy in the form of fats
Lipids provide insulation, e.g. birds and mammals can keep dry with a protective layer over fur or feathers
Lipids are the building blocks of many hormones and important in constructing cellular membranes
Lipids include fats, waxes, phospholipids, and steroids
Structure
A fat molecule consists of mainly glycerol and fatty acids
Glycerol is an organic compound (alcohol) with 3 carbons, 5 hydrogens, and 3 hydroxyl groups
Fatty acids have long chains of hydrocarbons attached to a carboxyl group
In a fat molecule, fatty acids are attached the glycerol’s carbons by dehydration synthesis with ester bonds through oxygen atoms
Ester bonds - a carbon with a single bond to a carbon, a double bond to an oxygen, and a single bond to an oxygen bonded to another carbon (R-O-C*--O *-R)
Fats are also called triacylglycerols/triglycerides because of their structure
They have “acyl” in them because the (O--C-C) group connecting the glycerol to the hydrocarbon chain is called an acyl group
Fatty acids are saturated if carbons have single bonds (“saturated” with hydrogen) while unsaturated fatty acids have double bonds
Oils
Unsaturated fats are typically liquid at room temperature and are called oils
One double bond in the molecule is monounsaturated and more than one double bond is polyunsaturated
Fats are only saturated when there are no double bonds
Unsaturated fats and oils contain cis fatty acids
Cis - hydrogens are in the same plane (causes a bend that prevents the acids from packing tightly, keeping them liquid at room temperature)
Trans - hydrogens are on two different planes
Trans Fats
Trans fats are artificially hydrogenated to make them semi-solid and have a more desirable consistency
Hydrogen gas is bubbled through oils which may double bonds from cis to trans
Omega Fatty Acids
Fatty acids not required but not synthesized by the human body are essential fatty acids and must be supplemented through the diet
The two known essential fatty acids are omega-3 and omega-6
omega-3 is polyunsaturated and the third carbon from the omega (w) carbon is connected to the fourth by a double bond
Good sources of omega-3 are fish like salmon and tuna
The furthest carbon away from the carboxyl group is numbered as the omega (w) carbon
Waxes
Made of long fatty acid chains esterified to long-chain alcohols
Because it’s hydrophobic, it covers some feathers and some leaves to prevent water from sticking to the surface
Phospholipids
Major constituents of the plasma membrane (outermost layer of living cells)
Composed of fatty acid chains attached to a glycerol or sphingosine backbone
Differs from fats because there are only two fatty acids forming a diacylglycerol, the third carbon is occupied by a modified phosphate group
A phosphate group attached to a diacylglycerol is a phosphatidate and becomes a phospholipid when modified by an alcohol
Phospholipids are amphipathic, meaning it has a hydrophilic and a hydrophobic part (the phosphate head is hydrophilic and the lipid tail contains the hydrophobic fatty acids)
phospholipids placed in water form a micelle, where the heads face the outside and the fatty acids face the interior
Steroids
Steroids have a fused ring structure
Hydrophobic and insoluble in water
Steroids all have four linked carbon rings and several have a short tail (like cholesterol)
Many steroids also have an -OH functional group, putting them in the alcohol classification
Most common steroid is cholesterol, which is mainly synthesized in the liver and is used for many things like testosterone and Vitamin D
Proteins
Proteins are long chains of different sequences of the 20 amino acids that each contain an amino group (-NH2), a carboxyl group (OH-C--O), and a variable group
Amino acids are linked by a peptide bond formed through a dehydration reaction; a long chain of amino acids is a polypeptide
Changes in temperature, pH, and exposure to chemicals may change the protein, making it lose its function (denaturation)
Types and functions
Proteins have the most diverse range of functions of all macromolecules and their structures vary greatly, although they are all polymers of amino acids in a linear sequence
Enzymes
Produced by living cells and are catalysts in biochemical reactions like digestion
Speeds up specific reactions by decreasing the amount of energy needed
Each enzyme is specific for the substrate (a reactant that binds to the enzyme) it acts on and may help in breakdown, rearrangement, or synthesis reactions
Enzymes that break down their substrate are catabolic, enzymes that build more complex molecules are anabolic, and enzymes that affect the rate of reaction are catalytic
All enzymes increase the rate of reaction and so are organic catalysts
Hormones
Chemical-signaling molecules, usually small proteins or steroids, secreted by endocrine cells that act to control specific processes
Primary functions
Digestive enzymes - catabolize nutrients into monomeric units
Transport - carry substances in the blood or lymph
Structural - make different structures e.g. cytoskeleton
Hormones - coordinate activities
Defense - protect from foreign pathogens
Contractile - effect muscle contraction
Storage - nourish the embryo/seedling during early development
Amino acids
Amino acids have a central alpha (a) carbon bonded to an amino group (NH2), a carboxyl group (COOH), a side chain, and a hydrogen atom
The side chain determines the nature of the amino acid (pH, polar/nonpolar)
Amino acids are represented by a single uppercase letter or a three-letter abbreviation (Valine - V or val)
Essential amino acids are necessary for proteins but not produced by the body
The sequence and number of amino acids determine the shape, size, and function of the protein
Amino acids are attached by a covalent bond known as a peptide bond, which is formed by a dehydration reaction
Linked amino acids form peptides, and multiple amino acids linked form a polypeptide with an amino group at one end (N or the amino terminal) and a carboxyl group at the other (C or carboxyl terminal)
After protein synthesis, proteins are modified through post-translational modifications that make it completely functional
The carboxyl group will lose its hydrogen proton and the amino group will have gained a hydrogen proton
A neutral molecule where parts have charges that balance each other out is called a zwitterion
Protein Structure
Enzymes bind to a specific substrate at a site known as the active site (region of enzyme surface where substrate binds)
Primary structure
The sequence of amino acids in a polypeptide chain is its primary structure
The sequence is determined by the gene encoding the protein; a change in the nucleotide sequence of the gene’s coding may affect protein structure and function
Secondary structure
The folding of the polypeptide in some regions is the secondary structure
Formed by hydrogen bonds between amino hydrogen and carboxyl oxygen on the backbones
The most common are the a-helix (spirally) and B-pleated sheet (zig-zaggy) structures, both of which are held by hydrogen bonds
Tertiary structure
The three-dimensional structure of a polypeptide is its tertiary structure
This structure is partly because of chemical interactions on the chains, primarily interactions between R groups
These interactions can counteract hydrogen bonds in standard secondary structures
Produced by hydrophobic interactions, ionic bonds, covalent bonds, and van der Waals forces
Quaternary structure
Interactions between subunits of polypeptides form the quaternary structure
Weak interactions between subunits stabilize the overall structure
Denaturation and protein folding
Temperature, pH, and chemicals may change the protein structure
When proteins lose their shape without losing their primary sequence, it is known as denaturation
Denaturation is often reversible because the primary structure of the polypeptide is conserved if the denaturing agent is removed
Chaperones
Protein helpers that assist during the folding process
They associate with the target protein and prevent aggregation of polypeptides that make up the complete protein structure
they disassociate from the protein after the process
Folding is critical to protein process
Nucleic Acids
DNA and RNA
Nucleic acids carry the genetic blueprint of a cell and instructions for cell functions
Two main types
Deoxyribonucleic acid (DNA)
Ribonucleic acid (RNA)
Genes
Genes may carry information to make protein or RNA products
Genome - the genetic content of a cell
Genomics - the study of genomes
DNA forms a complex with histone proteins to form chromatin - the substance of eukaryotic chromosomes, which may contain thousands of genes
DNA controls genes by turning them “on” or “off”
RNA
RNA is mostly involved with protein synthesis and regulation
DNA molecules never leave the nucleus, so messenger RNA (mRNA) are used to communicate with the rest of the sell
DNA and RNA
Made up of monomers known as nucleotides, which combine to form polynucleotides
Nucleotides have three parts
A nitrogenous base
Organic molecules (they contain nitrogen and carbon)
They are bases because they contain an amino group
Four possible bases
adenine (A) and guanine (G)
Classified as purines
Primary structure of a purine is two carbon-nitrogen rings
Cytosine (C), Uracil (U), and Thymine (T)
Pyrimidines
Primary structure is a single carbon-nitrogen ring
Uracil replaces Thymine in RNA; they are very similar, but thymine has a methyl group
A pentose sugar
Deoxyribose in DNA and ribose in RNA
On the second carbon (2’), ribose has a hydroxyl group and deoxyribose has a hydrogen
A phosphate group
Phosphate residue is attached to the hydroxyl groups of the 5’ carbon of one sugar and the 3’ carbon of the sugar of the next nucleotide, forming a 5’-3’ phosphodiester linkage
Not formed by dehydration reaction like others
Formed through the removal of two phosphate groups
DNA Double-Helix Structure
Sugars and phosphate lie on the outside of the helix, forming the DNA’s backbone
Nitrogenous bases are stacked on the interior in pairs; pairs are bound by hydrogen bonds
The two strands will run in opposite directions, making the 5’ carbon of one strand face the 3’ carbon of the other
This is called antiparallel orientation
It is important to DNA replication and nucleic acid interactions
Base complementary rule - A pairs with T and G pairs with C; this is due to the way the hydrogen bonds form between the nitrogenous bases
During DNA replication, each strand is copied, so the daughter DNA double helix contains one parental DNA strand and one newly synthesized strand
RNA
RNA is usually single-stranded and made of ribonucleotides linked by phosphodiester bonds
A ribonucleotide contains ribose, a nitrogenous base, and the phosphate group
Four main types of RNA
Messenger RNA (mRNA)
Carries messages from the DNA
Base sequence is complementary to the sequence of the DNA it was copied from, but U replaces T
Interacts with ribosomes and other machinery in the cytoplasm
Read in sets of three bases known as codons
Each codon codes for a single amino acid
This allows the mRNA to be read and produce a protein product
Ribosomal RNA (rRNA)
A major constituent of the ribosomes mRNA bind on
Ensures proper alignment of the mRNA and ribosomes
Catalyzes the formation of peptide bonds between two amino acids
Transfer RNA (tRNA)
One of the smallest of the four types
Carries the correct amino acid to the site of protein synthesis
The base pairing between tRNA and mRNA allows for the correct amino acid to be inserted into the polypeptide chain
tRNA has anticodons that pair with the codons on the mRNA
microRNA
Smallest RNA molecules
Regulate gene expression by interfering with the expression of certain mRNA messages
DNA and RNA comparison
Features of DNA and RNA |
|---|
Function |
Location |
Structure |
Sugar |
Pyrimidines |
Purines |
DNA dictates the structure of mRNA in a transcription process, and RNA dictates the structure of protein in a translation process
 Knowt
Knowt